Latest News and Press Releases
Want to stay updated on the latest news?
-
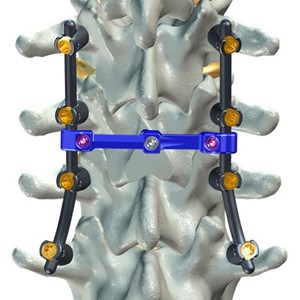
FLORENCE, S.C., Aug. 5, 2016 (GLOBE NEWSWIRE) -- DeGen Medical has received clearance from the FDA for its CONNECT-L™ Transverse Connector to be used with F1 MPS™ Modular Pedicle Screw...
-

FLORENCE, S.C., July 26, 2016 (GLOBE NEWSWIRE) -- DeGen Medical has received clearance from the FDA for its F1 MPS™ Modular Pedicle Screw System for JOUST™ Minimally Invasive Surgery (MIS)...
-

FLORENCE, S.C., Jan. 12, 2016 (GLOBE NEWSWIRE) -- DeGen Medical announced the launch of its cervical spine implant, Hyper-C™ Anterior Cervical Plate System. The Hyper-C™ system is an...
-

FLORENCE, S.C., Dec. 18, 2015 (GLOBE NEWSWIRE) -- DeGen Medical has received clearance from the FDA for our cervical interbody fusion device - Latitude-C™ Cervical Interbody Spacer System....
-

FLORENCE, S.C., Sept. 17, 2015 (GLOBE NEWSWIRE) -- DeGen Medical has received clearance from the FDA for its first cervical spine implant, Hyper-C™ Anterior Cervical Plate System. The...