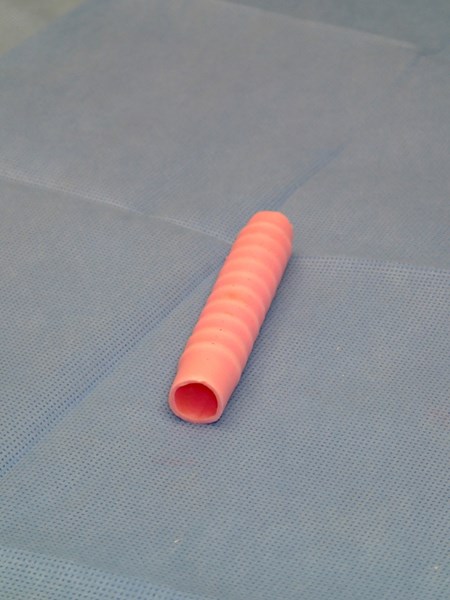
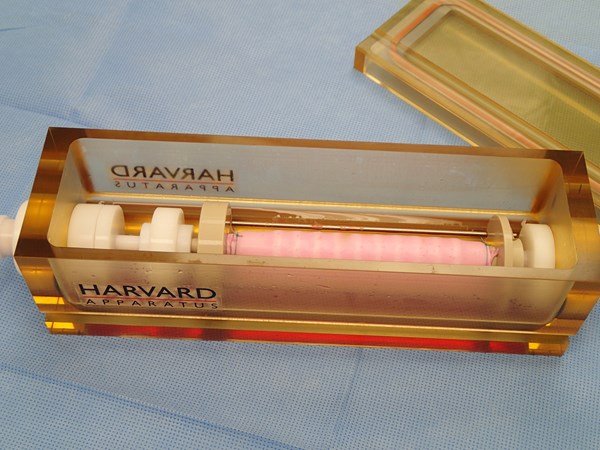
HOLLISTON, Mass., April 30, 2013 (GLOBE NEWSWIRE) -- Harvard Bioscience, Inc. (Nasdaq:HBIO), a global developer, manufacturer and marketer of a broad range of tools to advance life science research and regenerative medicine, announces that the "InBreath" tracheal scaffold and bioreactor system manufactured by Harvard Apparatus Regenerative Technology, Inc. (HART), its wholly owned regenerative medicine technology subsidiary, were used in the first successful transplant of a regenerated trachea in the United States. The recipient of the implant, two-year-old Hannah Genevieve Warren, is recovering at Children's Hospital of Illinois, where the surgery was performed on April 9, 2013. The surgery was also the world's first successful pediatric regenerated trachea transplant using a synthetic scaffold.
Photos accompanying this release are available at
http://www.globenewswire.com/newsroom/prs/?pkgid=18406
http://www.globenewswire.com/newsroom/prs/?pkgid=18407
Hannah was born on August 22, 2010 in Seoul, South Korea with tracheal agenesis (lack of a trachea), and was only able to breathe through a tube inserted in her esophagus that connected to her lungs. Tracheal agenesis is 100 percent fatal, and no child born with the condition has ever lived beyond six years. Hannah had lived in the intensive care unit for two and a half years at Seoul National Hospital before being transported to Illinois for the surgery.
This was the first regenerated trachea transplant surgery using a scaffold manufactured by HART and the implant used in the procedure was grown in one of HART's "InBreath" bioreactors. The scaffold and bioreactor were custom-made to Hannah's dimensions. The scaffold was seeded with bone marrow cells taken from the patient and incubated in the bioreactor for two days prior to implant. Because Hannah's own cells were used, her body has accepted the transplant without the use of immunosuppressive (anti-rejection) drugs.
The procedure was performed by a team led by Dr. Paolo Macchiarini of Karolinska University Hospital and Karolinska Institutet in Huddinge, Stockholm and Drs. Mark J. Holterman and Richard Pearl both of Children's Hospital of Illinois. The surgery was approved by the FDA under an Investigational New Drug application made by Dr. Holterman.
Dr. Mark Holterman, Professor of Surgery and Pediatrics at University of Illinois College of Medicine at Peoria, commented: "The success of this pediatric tracheal implantation would have been impossible without the Harvard Bioscience contribution. Their team of engineers applied their talent and experience to solve the difficult technical challenge of applying regenerative medicine principles in a small child."
David Green, President of Harvard Bioscience, commented: "We would like to congratulate Dr. Macchiarini, Dr. Holterman, Dr. Pearl and their colleagues for accomplishing the world's first transplant of a regenerated trachea in a child using a synthetic scaffold and giving Hannah a chance at a normal life. We also wish Hannah a full recovery and extend our best wishes to her family."
This surgery is the seventh successful implant of a regenerated trachea in a human using HART technology. Prior successes included the first ever successful regenerated trachea transplant in 2008, the first successful regenerated trachea transplant using a synthetic scaffold in 2011 and the commencement of the first clinical trial of regenerated tracheas in 2012. HART intends to begin discussions with the FDA and EU regulatory authorities in the near future regarding the clinical pathway necessary to bring this new therapeutic approach to a wider range of patients in need of a trachea transplant.
A subsequent press release will be issued to provide a link to an archived video of this press conference.
Video footage and additional still images pertaining to the surgery are available for media upon request.
About Harvard Bioscience
Harvard Bioscience, or HBIO, is a global developer, manufacturer and marketer of a broad range of specialized products, primarily apparatus and scientific instruments, used to advance life science research and regenerative medicine. HBIO sells its products to thousands of researchers in over 100 countries primarily through its 850 page catalog (and various other specialty catalogs), its website, through distributors, including GE Healthcare, Thermo Fisher Scientific and VWR, and via our field sales organization. HBIO has sales and manufacturing operations in the United States, the United Kingdom, Germany, Sweden and Spain with additional facilities in France and Canada. For more information, please visit www.harvardbioscience.com.
The Harvard Bioscience, Inc. logo is available at http://www.globenewswire.com/newsroom/prs/?pkgid=6426
About Harvard Apparatus Regenerative Technology, Inc.
Harvard Apparatus Regenerative Technology, Inc., a wholly owned subsidiary of Harvard Bioscience, develops and manufactures devices for use within the emerging field of regenerative medicine. These devices include bioreactors and synthetic scaffolds for growing tissue and organs outside the body; and injectors for stem cell therapy.
Registration Statement of Harvard Apparatus Regenerative Technology, Inc.
A registration statement including a prospectus relating to the common stock of Harvard Apparatus Regenerative Technology, Inc. has been filed with the SEC but has not yet become effective. These securities may not be sold nor may offers to buy these securities be accepted before the time the registration statement becomes effective. This press release shall not constitute an offer to sell or a solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or jurisdiction in which such an offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction.
Forward-Looking Statements
Some of the statements in this press release are "forward-looking" and are made pursuant to the safe harbor provision of the Private Securities Litigation Reform Act of 1995. These "forward-looking" statements include statements relating to, among other things, activities of Harvard Bioscience and the postponed IPO and other activities of Harvard Apparatus Regenerative Technology. These statements involve risks and uncertainties, including among other things, market conditions that may cause results to differ materially from the statements set forth in this press release. The forward-looking statements in this press release speak only as of the date of this press release. Harvard Bioscience expressly disclaims any obligation or undertaking to release publicly any updates or revisions to such statements to reflect any change in its expectations with regard thereto or any changes in the events, conditions or circumstances on which any such statement is based.
Photos are also available at Newscom, www.newscom.com, and via AP PhotoExpress.