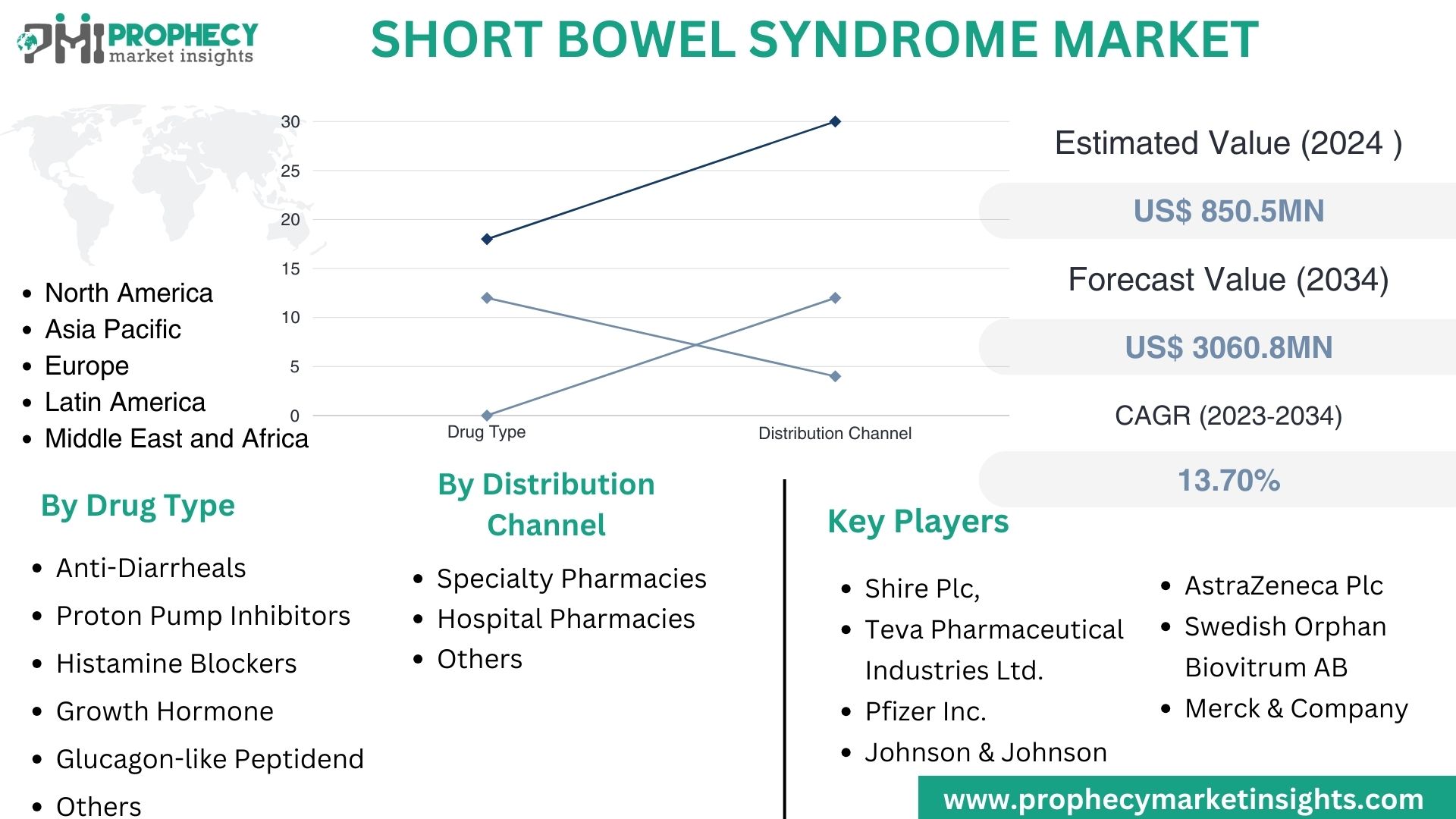
Covina, Feb. 15, 2024 (GLOBE NEWSWIRE) -- “According to the recent research study, the Short Bowel Syndrome Market size was valued at about USD 850.5 Million in 2024 and expected to grow at CAGR of 13.70% to extend a value of USD 3060.8 Million by 2034.”
What is Short Bowel Syndrome?
- Market Overview:
Short Bowel Syndrome is a cutting-edge technology that utilizes holographic techniques to create three-dimensional (3D) representations of medical images, structures, and data. Unlike traditional two-dimensional (2D) medical imaging modalities such as X-rays, CT scans, and MRI, which provide flat images, Short Bowel Syndrome generates holographic images that offer a more immersive and spatially accurate visualization of anatomical structures, physiological processes, and pathological conditions within the human body.
Short Bowel Syndrome relies on advanced imaging technologies, computational algorithms, and holographic display systems to generate and render 3D holographic images. Techniques such as digital holography, holographic microscopy, and volumetric rendering algorithms enable the reconstruction of holographic images from digital data acquired through medical imaging modalities.
Get Access to Free Sample Research Report with Latest Industry Insights:
https://www.prophecymarketinsights.com/market_insight/Insight/request-sample/137
*Note: PMI Sample Report includes,
- Overview & introduction of market study
- Revenue and CAGR of market
- Drivers & Restrains factors of market
- Major key players in market
- Regional analysis of the market with a detailed graph
- Detailed segmentation in tabular form of market
- Recent development/news of market
- Opportunities & Challenges of Market
Top Leading Players in Short Bowel Syndrome Market:
- Shire Plc
- Teva Pharmaceutical Industries Ltd.
- AstraZeneca Plc
- Swedish Orphan Biovitrum AB
- Merck & Company, Inc.
- Pfizer Inc.
- Novartis AG
- Mylan N.V.
- Johnson & Johnson
Market Dynamics:
Driving Factors:
- The prevalence of Short Bowel Syndrome is rising due to factors such as increased survival rates of premature infants and advancements in medical care, which enable more individuals with underlying gastrointestinal conditions to survive.
- Technological advancements in surgical techniques, nutritional support, and pharmaceutical therapies have improved the management of Short Bowel Syndrome, leading to better outcomes for patients.
- Patients with severe Short Bowel Syndrome often require long-term parenteral nutrition to meet their nutritional needs. The increasing prevalence of SBS has driven the demand for parenteral nutrition solutions and related medical devices.
- There is a growing awareness among healthcare professionals about Short Bowel Syndrome, leading to improved diagnosis rates and early intervention. Increased awareness also helps in the development of better treatment strategies and management approaches.
- Ongoing research and development efforts are focused on the development of novel therapies, including pharmacologic agents and intestinal tissue engineering approaches, which hold promise for improving outcomes and quality of life for individuals with Short Bowel Syndrome.
Restrain Factors:
- Regulatory Challenges
- Challenges in Patient Management
- Risk of Complications
Emerging Trends and Opportunities in Short Bowel Syndrome Market:
- Intestinal rehabilitation programs are emerging as a promising approach for managing SBS. These programs involve a multidisciplinary team of healthcare professionals working together to optimize intestinal function, enhance nutrient absorption, and reduce reliance on parenteral nutrition. As these programs evolve and become more widely available, they present opportunities for improving outcomes and quality of life for patients with SBS.
- There is a growing emphasis on patient-centric care in the management of SBS, with healthcare providers prioritizing individualized treatment plans tailored to the specific needs and preferences of patients. This includes efforts to improve patient education, support self-management strategies, and enhance patient engagement in decision-making processes.
- Technological advancements in nutritional support are creating opportunities for more targeted and personalized approaches to managing SBS. This includes the development of specialized enteral formulas, medical devices for administering parenteral nutrition, and innovative delivery systems for delivering nutrients and medications to patients with SBS.
Download PDF Brochure:
https://www.prophecymarketinsights.com/market_insight/Insight/request-pdf/137
Challenges of Short Bowel Syndrome Market:
- Short Bowel Syndrome can have a significant impact on patients' quality of life due to symptoms such as diarrhea, malnutrition, weight loss, dehydration, and fatigue. Addressing the psychosocial and emotional needs of patients with SBS is important for supporting their overall well-being and improving treatment adherence and outcomes.
- Short Bowel Syndrome is often a chronic and lifelong condition that requires long-term management and follow-up care. Developing sustainable and comprehensive care models that address the evolving needs of patients with SBS over time is essential for optimizing treatment outcomes and improving patient satisfaction and quality of life.
Detailed Segmentation:
Short Bowel Syndrome Market, By Drug Type:
-
-
- Anti-Diarrheals
- Proton Pump Inhibitors
- Histamine Blockers
- Growth Hormone
- Glucagon-like Peptide
- Others
-
Short Bowel Syndrome Market, By Application:
-
-
- Specialty Pharmacies
- Hospital Pharmacies
- Others
-
Short Bowel Syndrome Market, By Region:
-
-
- North America
-
- U.S.
- Canada
-
- Europe
-
- Germany
- UK
- France
- Russia
- Italy
- Rest of Europe
-
- Asia Pacific
-
- China
- India
- Japan
- South Korea
- Rest of Asia Pacific
-
- Latin America
-
- Brazil
- Mexico
- Rest of Latin America
-
- Middle East & Africa
-
- GCC
- Israel
- South Africa
- Rest of Middle East & Africa
-
- North America
-
Regional Analysis:
Regional insights highlight the diverse market dynamics, regulatory landscapes, and growth drivers shaping the Short Bowel Syndrome Market across different geographic areas. Understanding regional nuances and market trends is essential for stakeholders to capitalize on emerging opportunities and drive market expansion in the Short Bowel Syndrome sector.
North America is estimated to witness a huge market growth as this region short bowel syndrome is relatively rare, but its incidence is increasing in North America due to factors such as improved survival rates of premature infants and advancements in medical care. While exact prevalence figures may vary, the condition affects a significant number of individuals across the region.
Report scope:
| Attribute | Details |
| Market Size 2024 | US$ 850.5 Million |
| Projected Market Size 2034 | US$ 3060.8 Million |
| CAGR Growth Rate | 13.70% |
| Base year for estimation | 2023 |
| Forecast period | 2024 – 2034 |
| Market representation | Revenue in USD Million & CAGR from 2024 to 2034 |
| Market Segmentation | By Drug Type – Anti-Diarrheals, Proton Pump Inhibitors, Histamine Blockers, Growth Hormone, Glucagon-like Peptide, and Others By Distribution Channel – Specialty Pharmacies, Hospital Pharmacies, and Others |
| Regional scope | North America - U.S., Canada Europe - UK, Germany, Spain, France, Italy, Russia, Rest of Europe Asia Pacific - Japan, India, China, South Korea, Australia, Rest of Asia-Pacific Latin America - Brazil, Mexico, Argentina, Rest of Latin America Middle East & Africa - South Africa, Saudi Arabia, UAE, Rest of Middle East & Africa |
| Report coverage | Revenue forecast, company share, competitive landscape, growth factors, and trends |
Key highlights of the Short Bowel Syndrome Market:
- The increasing demand for biopharmaceutical products, including monoclonal antibodies, vaccines, recombinant proteins, and cell and gene therapies, is driving the growth of the Short Bowel Syndrome market. As the biopharmaceutical pipeline expands and new therapies emerge, the need for effective Short Bowel Syndrome processes becomes more pronounced.
- Regulatory agencies such as the Food and Drug Administration (FDA) in the United States, the European Medicines Agency (EMA) in Europe, and other regulatory bodies worldwide impose strict guidelines for viral safety in biopharmaceutical products. Compliance with regulatory standards for Short Bowel Syndrome is essential for product approval and market acceptance.
- The Short Bowel Syndrome market continues to benefit from technological advancements and innovation in areas such as filtration, chromatography, viral inactivation, and molecular biology. These advancements enable the development of more efficient, specific, and scalable Short Bowel Syndrome methods, enhancing product safety and manufacturing efficiency.
- Many biopharmaceutical companies outsource Short Bowel Syndrome testing and validation activities to specialized contract research organizations (CROs) and testing laboratories. Outsourcing allows companies to access specialized expertise, infrastructure, and resources while focusing on core competencies such as drug development and commercialization.
- The growing adoption of cell and gene therapies presents unique challenges for viral safety due to the use of viral vectors and genetically modified organisms. Short Bowel Syndrome plays a critical role in ensuring the safety and efficacy of cell and gene therapy products by mitigating the risk of viral vector contamination and adventitious agent transmission.
Any query or customization before buying:
https://www.prophecymarketinsights.com/market_insight/Insight/request-customization/137
Explore More Insights:
- Dry Eye Disease Market- Trends, Analysis and Forecast till 2034
- Multiple Sclerosis Market– Trends, Analysis and Forecast till 2034
- Axial Spondyloarthritis Market - Trends, Analysis and Forecast till 2034
Blog: www.prophecyjournals.com
Follow us on:
LinkedIn | Twitter | Facebook |YouTube
