Source: Precedence Research
Digital Therapeutics Revolution Transforming Chronic Care with Technology-Driven Solutions
According to Precedence Research, the digital therapeutics (DTx) industry is rapidly transforming the healthcare landscape, offering new, technology-driven solutions for the management of chronic conditions, mental health issues, and more.
Ottawa, Feb. 05, 2026 (GLOBE NEWSWIRE) -- Digital therapeutics are changing the way chronic conditions are managed, offering new opportunities for patient care and healthcare providers alike. According to data provided by Precedence Research, DTx solutions are not only receiving increasing regulatory approval but are also demonstrating real-world effectiveness through clinical trials and real-world applications. This growing trend highlights the potential for digital therapeutics to become a central element of future healthcare strategies.
The digital therapeutics (DTx) sector is quickly transforming healthcare, providing patients with tools to manage chronic diseases, mental health conditions, and other health issues through technology-driven solutions. From digital programs to mobile apps, DTx is integrating more seamlessly into everyday healthcare.
Invest in Our Premium Strategic Solution: https://www.precedenceresearch.com/request-consultation/34
Key Trends Driving Digital Therapeutics
Digital therapeutics encompass a wide range of technology-based solutions designed to treat or manage medical conditions through software, mobile apps, and other digital platforms. These solutions offer a personalized, patient-centric approach to managing conditions such as chronic diseases, mental health disorders, and metabolic health. The industry’s growth is fueled by several key factors:
- Increasing Regulatory Support:
Digital therapeutics are receiving growing recognition from regulatory bodies such as the FDA, which has cleared over 130 DTx products for use in the U.S. alone by 2022. Globally, several countries, including China, Germany, and the U.K., are following suit, providing clearer pathways for approval and reimbursement. This increasing regulatory support underlines the sector's growing legitimacy in the healthcare space. - Real-World Effectiveness:
Digital therapeutics have proven to be effective in real-world applications, with numerous companies reporting success in managing chronic conditions and improving patient outcomes. DTx solutions such as telehealth platforms, virtual physical therapy, and mental health apps are helping patients manage diseases such as diabetes, hypertension, and behavioral health disorders. - Innovative Technology Solutions:
Advancements in AI, machine learning, and data analytics are making digital therapeutics more effective. These innovations allow for personalized care by offering real-time tracking of patient health data, providing insights that can guide treatment adjustments and improve long-term health outcomes.
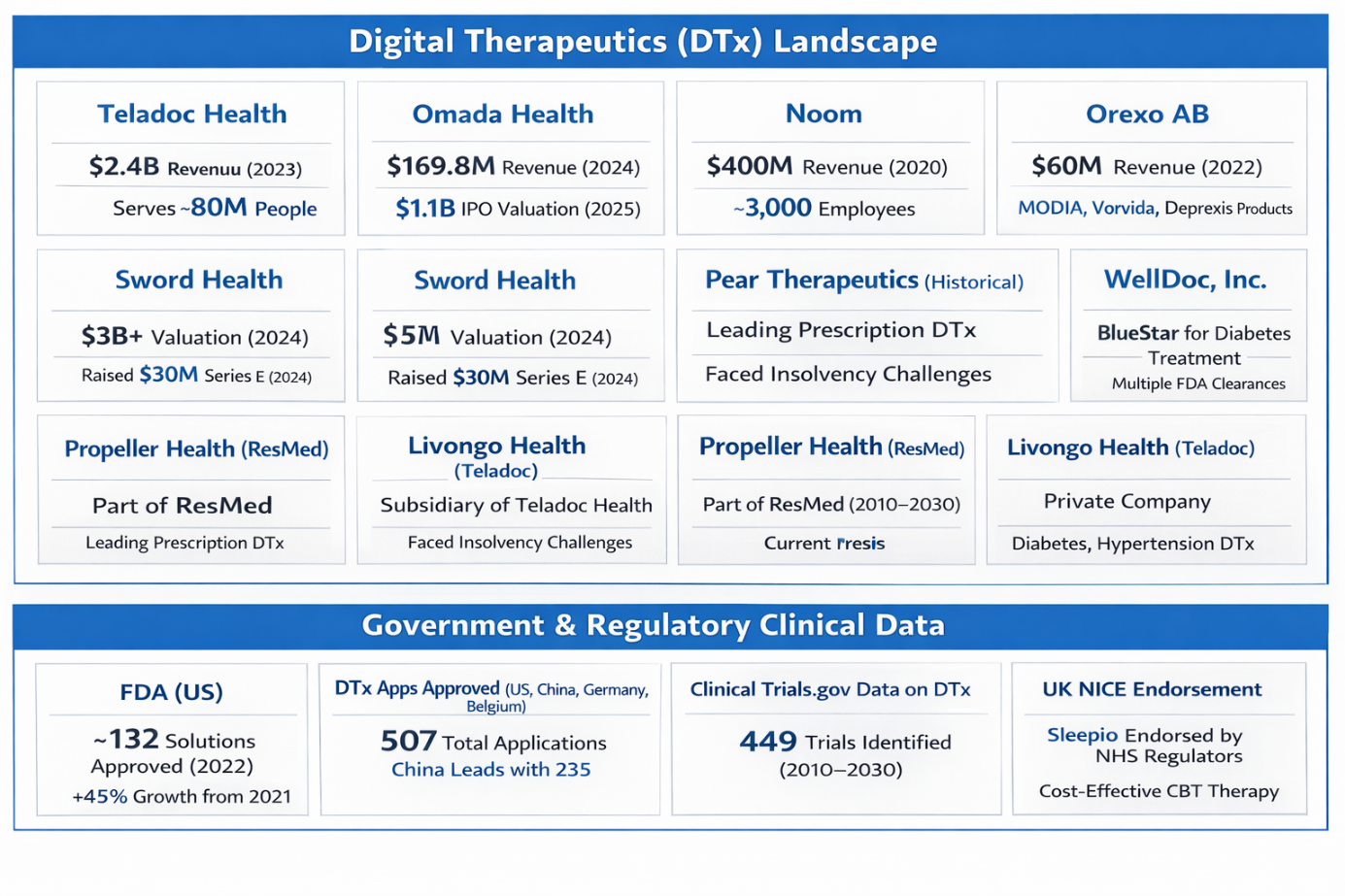
Key Players in the Digital Therapeutics Landscape
Several companies are leading the charge in the digital therapeutics space, developing cutting-edge solutions that are improving patient care worldwide. These companies are not just consumers of digital health technologies but are actively contributing to the innovation and expansion of the market.
- Teladoc Health:
A leader in telehealth services, Teladoc Health offers a broad range of digital solutions, including chronic care programs and mental health platforms. Its focus on accessible virtual care has positioned the company at the forefront of the digital health revolution, providing tools to help patients manage a variety of conditions remotely. - Omada Health:
Specializing in chronic disease prevention and management, Omada Health uses digital programs to address conditions like prediabetes, obesity, and hypertension. Through evidence-based digital interventions, the company is helping individuals take control of their health, preventing the onset of chronic diseases and improving long-term wellness. - Noom:
Known for its mobile app for weight management and behavioral health, Noom combines cognitive behavioral therapy with personalized coaching to help individuals achieve healthier lifestyles. The app focuses on behavior modification, providing users with the tools they need to make sustainable lifestyle changes. - Sword Health:
Sword Health is revolutionizing the physical therapy space with its AI-powered virtual physical therapy programs. These digital solutions are transforming musculoskeletal care by providing patients with personalized recovery programs that they can complete from the comfort of their homes. - Orexo AB:
A leader in the behavioral health space, Orexo AB offers a range of digital therapeutics solutions, including its MODIA and Deprexis platforms, which are designed to treat mental health conditions such as depression and anxiety.
These companies, among others, are pushing the boundaries of what is possible with digital therapeutics, each focusing on different areas of patient care and disease management. The continued growth and innovation in the sector will further cement digital therapeutics as a cornerstone of modern healthcare.
The Regulatory Landscape: Accelerating Approvals and Supporting Adoption
The regulatory approval process for digital therapeutics has been accelerating, allowing for broader adoption of these solutions. The U.S. FDA has approved a significant number of DTx products in recent years, and other regulatory agencies, such as the European Medicines Agency (EMA) and China's National Medical Products Administration (NMPA), are also increasingly recognizing the potential of digital therapeutics.
Here’s a look at the evolving regulatory landscape:
- FDA (U.S.):
The FDA has cleared over 130 digital therapeutics devices as of 2022. This reflects the agency’s growing confidence in the safety and efficacy of software-based therapeutic solutions. - Global Approvals:
Across the globe, there have been 507 DTx applications approved, with significant numbers coming from the U.S. (192), China (235), and Germany (55). This reflects the broadening global acceptance of digital therapeutics. - Clinical Trials:
The sector is witnessing significant investment in clinical research, with 449 clinical trials dedicated to digital therapeutics (DTx) registered between 2010 and 2030. This research underscores the effectiveness and viability of DTx solutions in treating chronic conditions. - UK NICE:
The UK’s National Institute for Health and Care Excellence (NICE) has endorsed DTx solutions like Sleepio, a digital therapeutic for cognitive behavioral therapy (CBT) in the treatment of insomnia, marking a significant milestone in the formal recognition of digital health technologies.
Regulatory Landscape: How Digital Therapeutics Are Being Approved
The regulatory approval process for digital therapeutics has been accelerating, as seen with increasing numbers of FDA approvals for software-based therapeutic devices. Here’s a snapshot of the regulatory landscape:
| Entity | Statistic | Explanation |
| FDA (US) | ~132 digital therapeutics approved (2022) | A significant rise in FDA-approved DTx solutions, reflecting growing adoption. |
| Global Approvals | 507 DTx applications approved (US: 192, China: 235, Germany: 55, Belgium: 25) | Demonstrates global acceptance and the rise of DTx in multiple regions. |
| Clinical Trials | 449 DTx clinical trials registered (2010-2030) | Shows the ongoing research and clinical validation efforts for DTx products. |
| UK NICE | Endorsed Sleepio as a cost-effective DTx for CBT | The first DTx to receive official endorsement by the UK’s NHS regulators. |
This data highlights the growing integration of digital therapeutics into mainstream healthcare. Regulatory bodies worldwide are acknowledging the potential of digital therapeutics to manage chronic conditions, providing clearer pathways for approval and reimbursement.
Key Insights
- Regulatory Growth: The approval of digital therapeutics is rapidly increasing, with more than 130 solutions cleared by the FDA alone in 2022. This demonstrates how the sector is becoming more widely accepted by regulators.
- Growing Adoption: Companies like Teladoc Health and Omada Health are generating substantial revenue from digital therapeutics, showcasing real-world application and acceptance in chronic disease management.
- Clinical Research: With 449 clinical trials focused on DTx products, this sector continues to be a critical area of research, validating the effectiveness of digital solutions in healthcare.
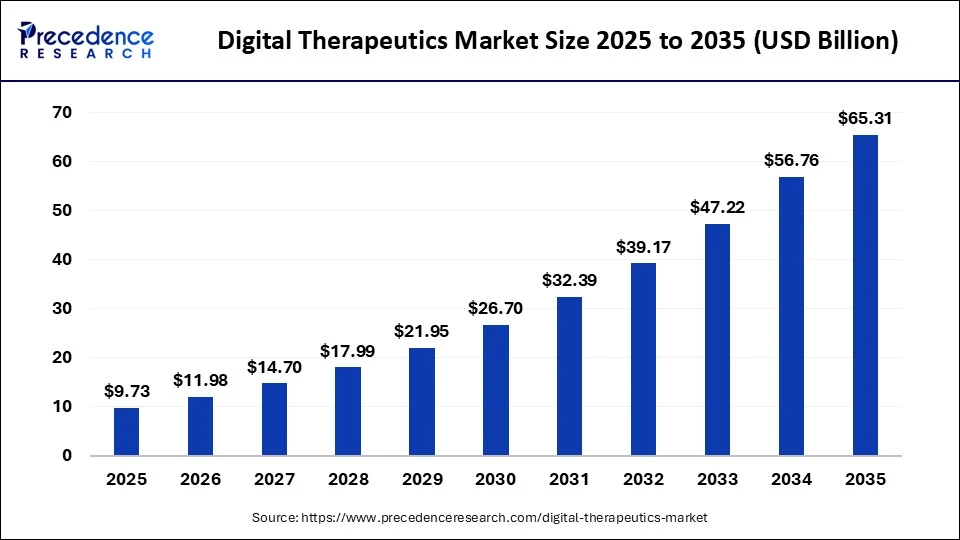
According to Precedence Research, the global digital therapeutics market size is valued at USD 9.73 billion in 2025 and is projected to reach around USD 56.76 billion by 2034, growing at a CAGR of 21.65%. This growth is driven by the rising chronic disease burden, expanding reimbursement pathways, and rapid adoption of clinically validated digital care platforms.
Conclusion: The Future of Digital Therapeutics
The digital therapeutics industry is experiencing unprecedented growth, with increasing regulatory support and real-world evidence of effectiveness. Companies are innovating rapidly, and new solutions are continuously emerging to address a wide range of health conditions. As the sector expands, these technologies will likely become integral to healthcare strategies, transforming the way chronic conditions are managed and offering better, more accessible care for patients worldwide.
Request Research Report Built Around Your Goals: sales@precedenceresearch.com
About Us: Precedence Research
Our Legacy: Rooted in Research, Focused on the Future
Looking for research that drives real results? Precedence Research delivers strategic, actionable insights, not just data and charts. Based in Canada and India, our team specializes in customized market analysis, executive-level consulting, and tailored research solutions that go beyond traditional survey methodologies to support business growth with precision and confidence.
Insight-Driven
We turn complex data into clear, strategic insights that power confident business decisions.
Innovation-Led
We continuously refine our methods to stay ahead of trends and emerging market forces.
Industry-Agnostic
From tech to healthcare, we serve clients across sectors with tailored, actionable intelligence.
Customer-Centric, Future-Focused, Result-Oriented
We work as strategic partners, engaging deeply with clients to co-create impactful solutions.
Our Commitment: Delivering Intelligence That Drives Transformational Growth
What do we do? We turn data noise into clarity. Through sharp research, agile thinking, and tech-enabled tools, we fuel brands, disrupt markets, and lead with insight that drives unstoppable growth.
Contact Us
USA: +1 8044 419344
APAC: +61 4859 81310 or +91 87933 22019 or +6531051271
Europe: +44 7383 092 044
Email: sales@precedenceresearch.com
