BOSTON, May 07, 2026 (GLOBE NEWSWIRE) -- Sesen, a company specializing exclusively in life sciences translation, multilingual content operations, and regulated language workflows, today announced the launch of the Sesen Labeling Workspace, a centralized environment designed to help pharmaceutical, biotechnology, medical device, CRO, and healthcare organizations manage global labeling content, translation, and review workflows with greater control, consistency, and traceability.
Powered by SesenGPT, Sesen’s hybrid translation system for regulated content, the Labeling Workspace brings together AI-assisted translation, professional native medical linguist review, terminology management, translation memory, validation workflows, and audit-ready documentation within a structured environment built for global life sciences labeling programs.
Global labeling translation remains one of the most complex areas of multilingual content management. Regulatory, labeling, localization, medical, and quality teams must coordinate recurring updates across markets while maintaining consistency across terminology, formatting, version history, and submission-related documentation. Traditional workflows often rely on disconnected emails, spreadsheets, file transfers, and manual review cycles, increasing the risk of delays, inconsistencies, and limited visibility across multilingual content operations.
The Sesen Labeling Workspace is designed to address these challenges by centralizing global labeling management, translation, validation, review, approval, and delivery processes within a controlled workflow environment. The workspace also provides centralized collaboration for regulatory, labeling, localization, medical, and quality stakeholders involved in multilingual review and approval processes. The platform supports drug labeling lifecycle updates, Instructions for Use (IFU) translations, safety updates, CCDS and SmPC revisions, packaging content, post-approval labeling changes, and multilingual regulatory submission materials.
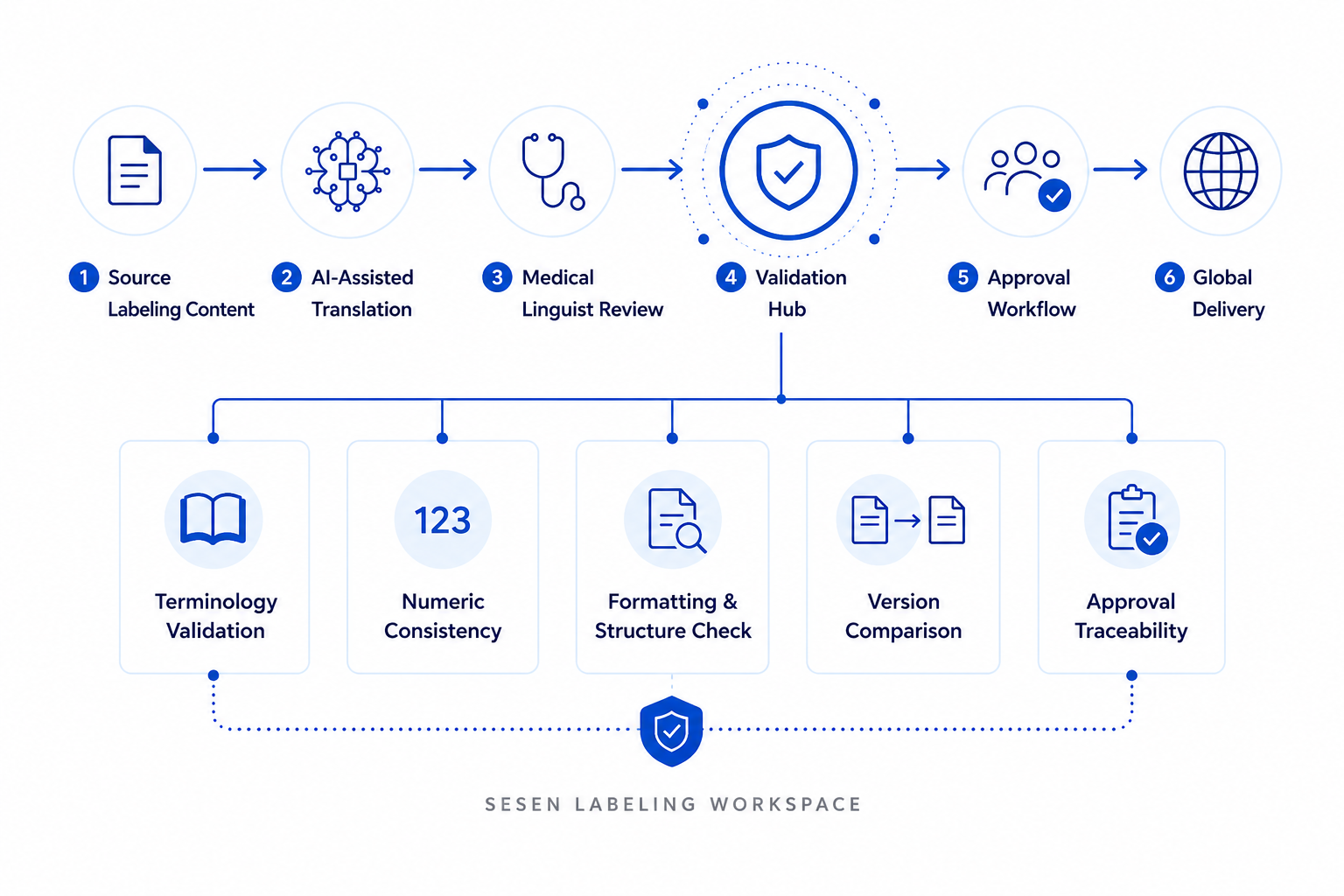
At the core of the workspace is the Validation Hub, designed to support AI-driven governance for multilingual regulated content. The Validation Hub provides terminology validation against approved glossaries, numeric consistency checks, formatting and structural verification, version comparison, review logs, and approval traceability. This structured validation process helps life sciences organizations reduce manual QA effort while supporting submission-ready multilingual content with end-to-end traceability, version visibility, and audit-ready documentation.
“The life sciences industry is looking for practical ways to use AI without losing the control required for regulated content,” said Shunee Yee, CEO of Sesen. “The Sesen Labeling Workspace brings AI-assisted translation, expert human review, and validation workflows together in a structured environment designed for real-world labeling operations. Our goal is to help clients improve speed and consistency while maintaining the oversight, traceability, consistency, and quality required for regulated global content.”
Unlike standalone AI translation tools, SesenGPT operates within a controlled hybrid workflow that integrates translation memory, approved terminology, automated QA checks, and professional medical linguist review. The workspace intelligently applies AI-assisted translation to new or modified content while leveraging previously approved multilingual assets and terminology to improve consistency across recurring labeling updates.
The Sesen Labeling Workspace supports translation programs across 150 languages and reflects Sesen’s broader commitment to helping life sciences organizations manage multilingual clinical, regulatory, labeling, and product content with greater operational efficiency and quality.
Sesen is also launching pilot programs that allow organizations to evaluate the Labeling Workspace using real-world labeling and regulatory content. Pilot programs are designed to help teams assess improvements in multilingual consistency, workflow visibility, turnaround time, and validation-driven review processes within regulated environments.
To learn more, visit:
www.sesen.com/life-sciences-labeling-workspace/
About Sesen
Sesen specializes exclusively in multilingual content operations for the life sciences industry. The company supports pharmaceutical, biotechnology, medical device, CRO, and healthcare organizations with AI-enabled translation workflows, regulatory labeling support, multilingual content management, and validation-driven language operations across 150 languages. Combining professional native medical linguists, terminology management, workflow automation, and ISO-certified quality systems, Sesen helps life sciences teams manage regulated global content with greater consistency, visibility, and operational efficiency.
Media Contact
Nikki Andrews
press@sesen.com
Website: www.sesen.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/0861b412-0627-4335-b00d-5d6d151e18cd
